 
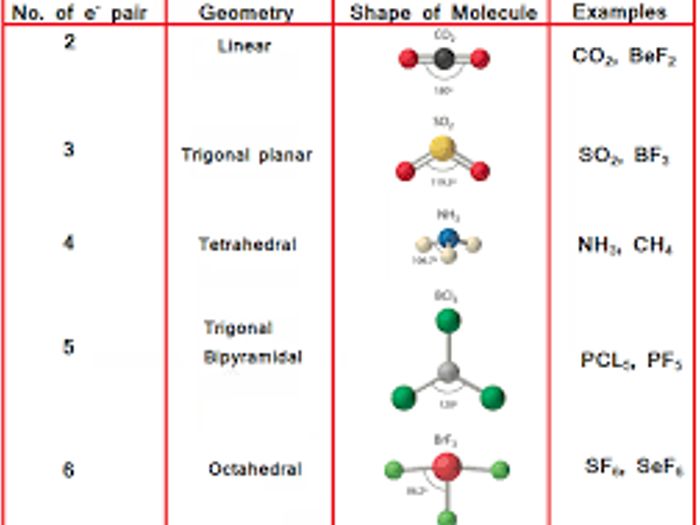
The VSEPR number helps in determining the shape of the molecule. 
This is useful in determining the bond angles. Step 5: Observe and find the lone pair-lone pair interaction, lone pair-bond pair interaction and bond pair-bond pair interaction. Step 4: Determine a stable arrangement of electron pairs around the central atom so that they experience the least repulsion. Step 3: Count the total number of bonded pairs and valence shell electrons by analyzing the atoms bonded with the central atom. Step 2: The least electronegative atom is taken as the central atom in the structure. Step 1: First, draw the lewis electron dot arrangement for a given ion or molecule. Steps to Use The VSEPR theory To Predict the Shape of Molecules
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
